Secrets of cells revealed by innovative droplet-based tools
A European bioengineer has pushed the boundaries of cell manipulation by pioneering a new, droplet-based method of study. His innovation, allowing for the highly detailed observation of cells, has resulted in the launch of a spin-off company as well as further research into cell cultures. Fuelled by a European Research Council (ERC) grant, the technology promises to open doors to new applications in pharmaceuticals, diagnostics and medical research.
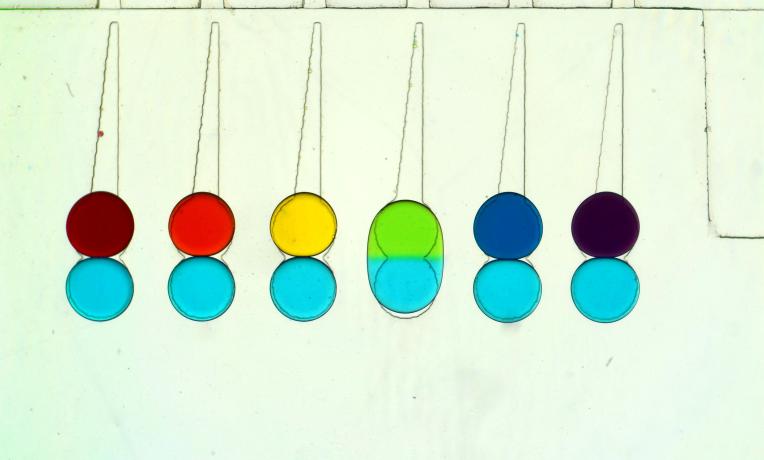
Dr Charles Baroud, Head of the Pasteur-Polytechnique joint group on Physical Microfluidics & Bio-engineering, France, has developed a microfluidic droplet-based technique that enables researchers to enhance cell manipulation. The technique, which allows for the observation, measurement and manipulation of individual cells at an unprecedented level, could unlock research in a vast array of disciplines and, as a result, has sparked several commercial opportunities.
The first spin-off company, Stilla Technologies, was launched in 2013, and continues to sell these microfluidic droplet-based tools around the world. “We quickly realised that our droplet tools were better than what was on the market,” explains Dr Baroud. “An ERC Proof of Concept grant, along with seed funding and venture capital, enabled us to scale this business up very quickly and the company now has about 60 staff.”
Continued interest about the practical applications from biologists has resulted in the formation of a second spin-off company, expected to be launched in early 2020. The focus of this business will be on cutting-edge 3D cell cultures, which can give researchers an incredibly clear picture of what is happening in the body. Both a second ERC Proof of Concept grant and public funding from France have been secured, and Dr Baroud and his team are currently looking for seed funding.
“We initially thought that we would develop techniques that could answer some fundamental biological questions by themselves,” he explains. “But what we actually found out was that we were developing tools that enable others to then develop more specific tools – we were just moving up a level.” As a result, the research could stimulate significant discoveries across disciplines.
For example, if researchers can see how cells are behaving, they may then want to know how this behaviour changes when a particular chemical is added. Dr Baroud’s innovations offer companies the means to develop the tools to answer this question.
Medical staff also have the opportunity to extract patient cells and see how they might respond to specific drugs. This could mark an important contribution to personalised medicine, as well as to sustainable drug production, by significantly increasing the ability of researchers to explore different conditions in a single experiment.
Dr Baroud leads a team comprised of biologists, engineers, physicists and chemists, and believes that this multidisciplinary approach has been key to the project’s success. “Before the ERC grant, my team and I worked solely on thermodynamics from a physics and engineering background, examining how water droplets in oil can be manipulated,” he says. “I was beginning to realise that the tools for manipulating droplets were insufficient in terms of having useful applications, so I wanted to integrate biology into our lab work.”
This, says Dr Baroud, would simply not have been possible without the ERC. “The grant also suited me very well because it enabled risk-taking,” he says. “Things didn’t always go exactly as planned but this was to be expected. And the fact is that the ERC never blocked anything. This, I think, was very important.”
Biography
Charles Baroud is professor of Mechanics at Ecole Polytechnique, where he founded the microfluidics research activity in 2002. Since 2018, he also heads a joint research unit at Institut Pasteur (Paris), working at the interface between engineering, physics, and biology. His work at Polytechnique initially focused on multiphase fluid flows in microfluidics, using a mixed experimental and theoretical approach. This work has led to new fundamental insights on drop and bubble dynamics in complex geometries, as well as to the invention of a unique toolbox for droplet production and manipulation. More recently, the research focus has shifted, thanks in part to an ERC starting grant, to applying the droplet methods to address problems in quantitative biology. Typical biological questions that are studied revolve around quantifying the variability between individual cells and on their interactions, and on the emergence of population-scale behavior as a result of this variability. Examples include studying immune-cancer interactions, the variability in response of bacteria to antibiotics, or building 3D micro-tissues to mimic in vivo biology. The early droplet microfluidics work has been transferred to a spinoff company, Stilla Technologies, while the cell interaction work is currently being incubatied in view of spinning off a second startup. Prior to his faculty position, Charles Baroud obtained an undergraduate degree at MIT and a PhD at the University of Texas, Austin, both at the interface between Physics and Mechanical Engineering, followed by a post-doc at Ecole Normale Superieure in Paris.