How is your brain powered?
The brain is one of the most energy-consuming organs. It represents only 2% of the weight of an adult but it uses 20% of the energy produced by the body. Efficient energy supply is crucial for the brain so that our memory, mobility and senses can function normally. On the occasion of the ‘Month of the Brain’ in May 2013, ERC Advanced grantee, Prof. David Attwell at University College London (UK) explains the mechanisms through which the brain is powered. Understanding these mechanisms may allow the development, in the long-term, of innovative therapies for cerebrovascular disorders.
Computers need a power supply to process information, for example when typing a document or surfing the web. The same goes for the operations performed by our brain cells. With his BRAINPOWER project, Prof. David Attwell is setting out to understand how and where the energy supply of the brain is controlled. “The brain is powered by the glucose and oxygen which are provided to it in the blood. Because nerve cells use lots of energy, when they are active they signal to nearby blood vessels, telling the vessels to dilate, in order to deliver more substrates for energy production. These are the mechanisms we are studying.”
Failure of the energy supply to central nervous system tissue contributes to a wide range of neurological disorders such as stroke and spinal cord injury. These conditions have important social, economic and healthcare impact, as they are associated with increased disability and a larger risk of mortality, and they are rising in incidence in our increasingly ageing society.
It was long believed that cerebral blood flow is controlled by the large arterioles penetrating the brain from its surface. A few years ago, Prof. Attwell and his team discovered an alternative vascular mechanism that might control the brain’s blood flow, mediated by a specific type of contractile cell called ‘pericytes’.The team believes that these pericytes, situated on small capillary vessels, may play a crucial role in regulating the energy to brain nerve cells. “This hypothesis was controversial at that time but it is increasingly being accepted by the scientific community,” Prof. Attwell says. “Building on this initial discovery, there is now evidence that may help to explain some of the nerve cell damage that occurs after a stroke. When a blood clot occludes a cerebral artery, nerve cells are damaged, but even if the clot is removed with medicines a long-lasting decrease of blood flow remains, and this can damage more nerve cells. This may be caused by an abnormal constriction of pericytes triggered by the initial stroke."
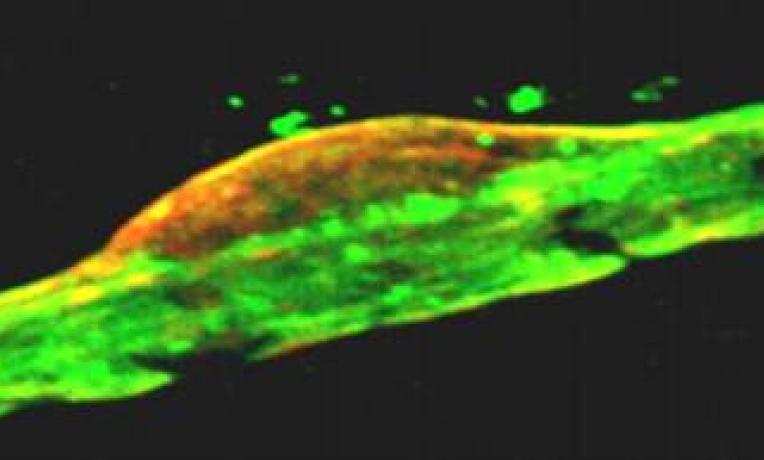
The research team uses an advanced imaging methodology to understand how these intriguing cells function. With a two-photon microscope, researchers can study pericytes in brain slices as well as in anaesthetized rats and mice, and determine what dilates or constricts them. To mimic cerebrovascular diseases such as stroke, the team is using brain slices superfused with reduced oxygen and glucose concentrations, as well as anaesthetised animals with experimentally reduced blood flow (in collaboration with the groups of Martin Lauritzen in Copenhagen and Alastair Buchan in Oxford). “This approach has allowed us to suggest that capillaries dilate before arterioles when nerve cells are active in normal conditions, and we are investigating whether pericytes constrict in response to stroke and then die, leaving the capillaries permanently occluded”, notes Prof. Attwell.
Understanding how pericytes behave may also lead to the development of new pharmacological treatments to prevent pericyte death, which could help improve the number of nerve cells surviving after a stroke. A (currently hypothetical) medicine could be injected at the same time as the blood clots are removed, and would help patients to recover a normal blood flow to their brain. Prof. Attwell says: “Cerebrovascular conditions are currently very hard to treat, so this research gives the promising perspective that we may be able, in the long-term, to save lives”.
“The ERC funding not only cuts down on paperwork compared to usual schemes, but it has also been crucial for my work as it has allowed me to employ talented researchers in my team ”, declares Prof. Attwell. “It has also provided us with the necessary equipment, i.e. the 2-photon microscope, which allows us to see deeper into brain slices and into the living brain than is possible with a normal microscope, and hence to advance our research in neuroscience”.

