Medical imaging: Can we predict how cancer patients will respond to nanotherapies?
Despite holding out promise for cancer treatments, nanomedicine lacks the means to predict patient response to tumour-targeting therapy. Prof. Twan Lammers has devised imaging which differentiates between those likely to benefit and those unlikely to do so.
A significant barrier for the nanopharmaceutical industry tackling cancer is that while some patients treated with nanomedicine show major improvements in survival rates and quality of life, many others do not respond well. This can result in delays in receiving appropriate treatment. A step forward would be the ability to differentiate likely responders from non-responders, prior to their selection for studies or treatment.
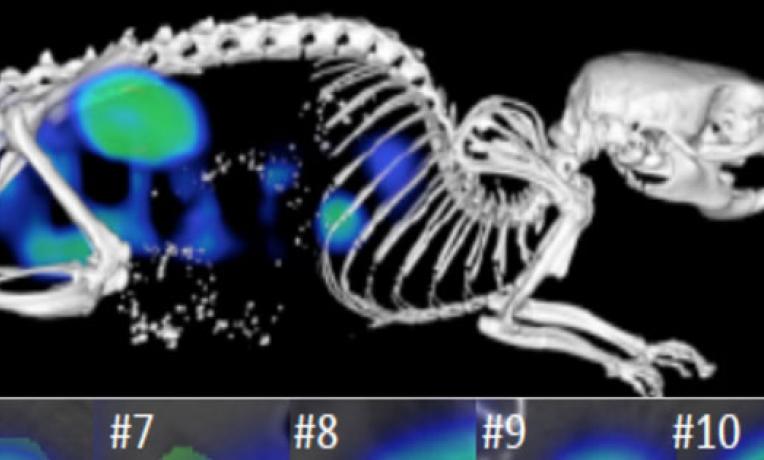
Prof. Lammers addressed this clinical gap through the development of nanomedicine imaging which uses (radio-)tracer labels to pre-select patients for inclusion in clinical trials.
Individual EPR profiles for each tumour
To work effectively, nanomedicines depend on the so-called Enhanced Permeability and Retention (EPR) effect, whereby molecules are thought to accumulate more readily in tumours than in normal tissue. However, the EPR effect is variable across patients and can even present differently within lesions in the same patient.
Unlike virtually all new anticancer drugs which have protocols to stratify potential responders from non-responders, nanomedicine tumour-targeting technology does not have the same safeguard.
Prof. Lammers was able to conduct non-invasive imaging of tracer-labelled nanocarriers that accumulate in tumours through the EPR effect. Patients evidencing a low degree of EPR, and so unlikely to respond, can then be excluded from treatment and referred for established or experimental interventions. Likewise, patients with a high level of EPR can expect relatively impactful treatment.
Funded by the ERC, his team applied both nanodiagnostic and nanotheranostic approaches. In the former case, drugs are precluded from the nanocarrier screening step. Whereas with the theranostic approach, both the drug and the imaging agent are present in the same formulation, providing valuable in situ information on target site accumulation of the nano-drug under investigation.
“Options are currently being explored, by us and others; not only in terms of making and upscaling the therapeutic application of nanoproducts, but also for defining the optimal biomarkers and measures to be used for patient pre-selection,” Prof. Lammers summarises. “These include things like the percentage of injected dose, distribution of dose within the tumour and the kinetics of probe accumulation.”
A key therapeutic tool for the future
To exploit the technology’s commercial potential for the pharmaceutical and diagnostics industry, the research team is working to customise the technique for integration with PET-MRI imaging.
“In the future, theranostic techniques are likely to be used to exploit nanomedicine more efficiently in the clinic, for immunotherapy purposes,” Prof. Lammers enthuses before adding, “We are currently exploring several options in this direction. Nanomedicine is still in its infancy, but with increasing discussion and success stories, I am convinced this will become a crucial therapeutic tool for cancer patients.”
This article was first published in the CORDIS Results Pack on Nanomedicines - innovative ways of treating challenging conditions
Twan Lammers obtained a DSc degree in Radiation Oncology from Heidelberg University (Germany) in 2008 and a PhD degree in Pharmaceutics from Utrecht University (Netherlands) in 2009. In 2014, he was appointed as Professor of Medicine at RWTH Aachen University Clinic (Germany). He is the Head of the Department of Nanomedicine and Theranostics, which is part of the Institute for Experimental Molecular Imaging. His primary research interests include image-guided drug delivery and tumour-targeted combination therapies.
This article was first published in the CORDIS Results pack on nano medicines.