Novel therapy starves the engine driving cancer cell growth
European researchers have identified a novel approach to prevent the growth of cancer tumours and inhibit them from spreading, potentially leading to highly effective treatments with fewer side effects.
The work, conducted in the ECMetabolism project with support from the European Research Council (ERC), builds on prior research into the formation of blood vessels – a process known as angiogenesis – that supply tumours with nutrients and oxygen. Blood vessels also provide a route for cancer to metastasise from the original tumour and spread elsewhere in the body.
Current anti-angiogenic therapies aim to destroy all tumour blood vessels and starve the cancer cells. But the treatment often provokes numerous side-effects, while its effectiveness can be inhibited by patient resistance and may even increase the risk of metastasis.
“There was an unmet need for novel anti-angiogenic strategies with fundamentally distinct mechanisms,” explains Peter Carmeliet, who led the ECMetabolism research at the VIB Vesalius Research Center of KU Leuven in Belgium. So the research team set out to develop a new anti-angiogenic concept targeting key metabolic pathways in endothelial cells which line the blood vessels, rather than the blood vessels themselves. “Our work has advanced the current state of the art and scientific understanding in the field of metabolism and angiogenesis.”
A little-known cell
When the ERC project began more than five years ago, very little was known about how endothelial cell metabolism regulates vessel sprouting, a process in which existing blood vessels grow offshoots that become new vessels. Carmeliet hypothesised that endothelial cell metabolism is the engine driving vessel sprouting and that turning down the engine in endothelial cells would provide an alternative anti-angiogenic treatment.
What is the key to turning down the engine? Glucose, more commonly known as blood sugar, which Carmeliet and his team identified as a major fuel source for endothelial cell metabolism through a process called glycolysis.
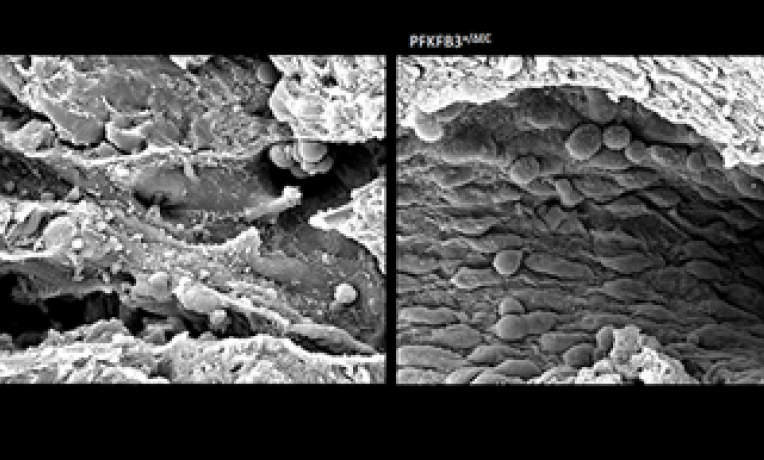
“We showed that endothelial cells are addicted to glycolysis, and that a partial and transient reduction of glycolysis by blocking the glycolytic activator PFKFB3 – using the commercially available small molecule compound, 3PO – inhibits pathological angiogenesis without systemic effects. To the best of our knowledge, these are the first findings showing that a metabolic pathway (glycolysis) can be a target for tumour vessel normalisation and anti-metastatic therapy,” Carmeliet says.
Crucially, in vitro and in vivo animal studies conducted by the ECMetabolism researchers showed that other cell types do not rely as much on glycolysis as endothelial cells and are therefore able to switch to alternative metabolic pathways, potentially resulting in far fewer side-effects for patients.
Following on from that ground-breaking research, Carmeliet and his team are now shifting focus towards translating the results into metabolic anti-angiogenic treatment candidates for new drugs, including screening a library of existing pharmaceutical compounds for an alternative PFKFB3 inhibitor that would be more active and easier to administer.
“The ERC grant gave us the freedom and trust to pursue innovative, high-risk/high-gain research at the frontline of life sciences, attracting a critical mass of researchers over a longer period of time and making significant progress that would not have been possible via other funding organisations,” Carmeliet says. “With this support we have been able to produce ground-breaking scientific insights and discoveries in an unexplored research field that will be of great benefit for the scientific community and clinical medicine.”