Decades-old chemistry mystery solved as elusive molecule is caught for the first time

Publishing in Science Advances, researchers from KTH Royal Institute of Technology in Stockholm and Kinetic Chemistry Research in Mountain View, California, say their discovery of long-theorised, oxygen-rich tetroxides has implications for several fields, including atmospheric chemistry, biochemistry and medicine and combustion chemistry.
'This compound is the equivalent of the Higgs boson for the oxidation chemistry community, says Barbara Noziere, professor of physical chemistry at KTH Royal Institute of Technology and ERC grantee. 'Its existence was assumed for decades but nobody had ever seen it.'
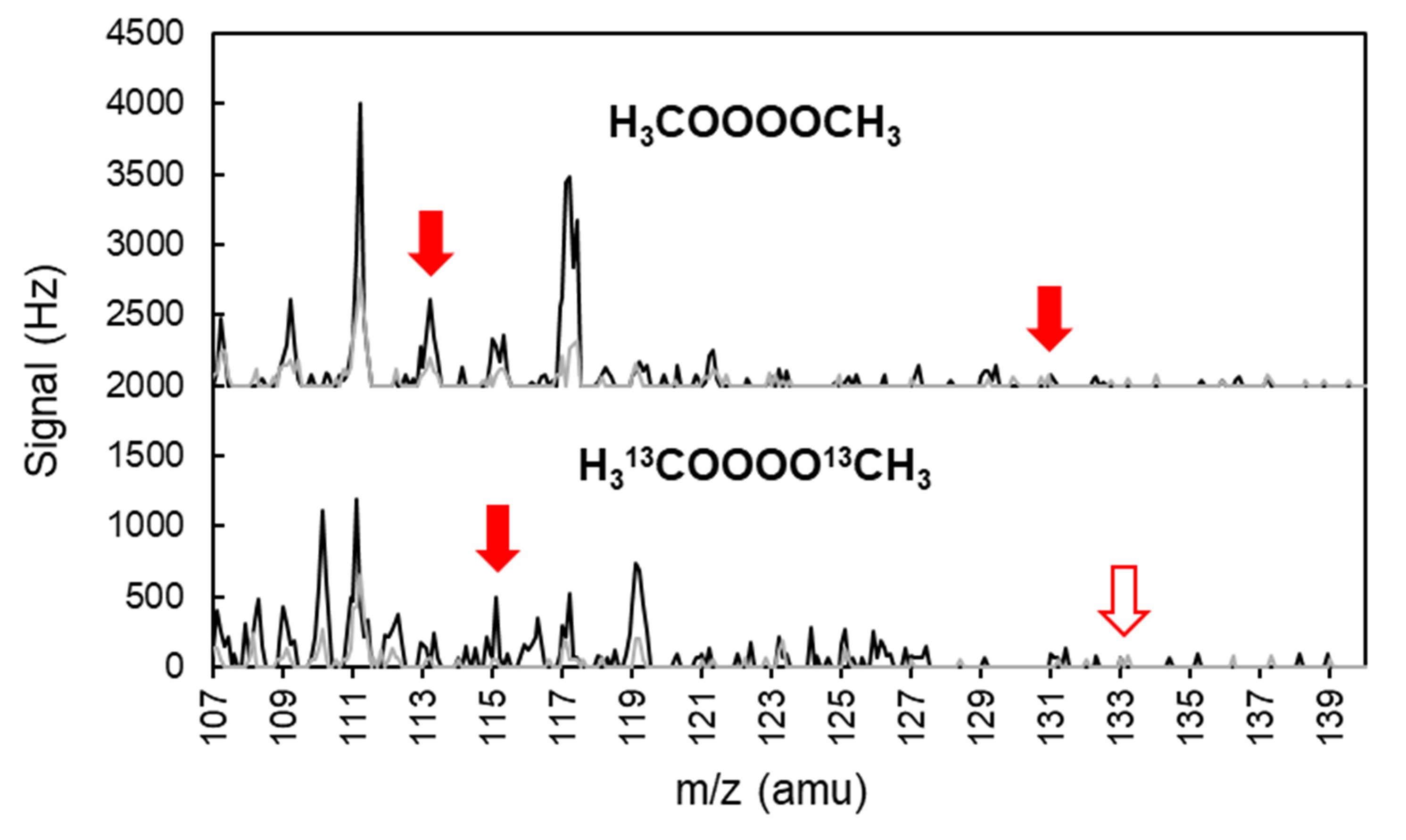
First theorised in the 1950s, tetroxides have been predicted to appear for a fleeting moment when two organic radicals react together, creating a molecule with four oxygen atoms in a row – a process called the Russell mechanism.
Although they disappear almost immediately, tetroxides play important roles in all the processes where organic compounds - or carbohydrates - are ‘burned’ in contact with air, such as in fires, candlelight flames, car engines, but also at low temperature in Earth’s atmosphere and inside living organisms.
Evidence of their existence until now had been indirect, contradictory or based on cold and extreme laboratory conditions. The team confirmed their presence using a unique mass spectrometric technique refined to detect highly unstable molecules without destroying them.
Professor Noziere says the findings also have significant implications for medical science, including research on oxidative stress and cancer therapies, where Russell mechanism is already being used in emerging therapeutic approaches.
Surprisingly, the researchers found that in air tetroxides are relatively stable, unlike in the conditions used in previous studies.
'The study confirms that tetroxides can exist at room temperature, in air, without needing extremely cold conditions used in earlier experiments,’ Noziere says.
The discovery that they can exist outdoors and inside living organisms means they can follow unexpected reaction steps and result in previously unexpected oxidation products that now need to be studied further.
This could influence how long pollutants – such as paint solvents or smoke – last in the atmosphere, the creation of other airborne compounds, or even of aerosol particles.
Noziere says that measuring their lifespan - between 0.2 and 200 milliseconds - also helps scientists understand how fast certain reactions move and what other products they can lead to.