Regenerating bones and teeth
ERC grantee Alvaro Mata, from Queen Mary’s School of Engineering and Materials Science, has developed a new way to grow mineralised materials which could regenerate hard tissues such as dental enamel and bone. The findings are reported today in Nature Communications.
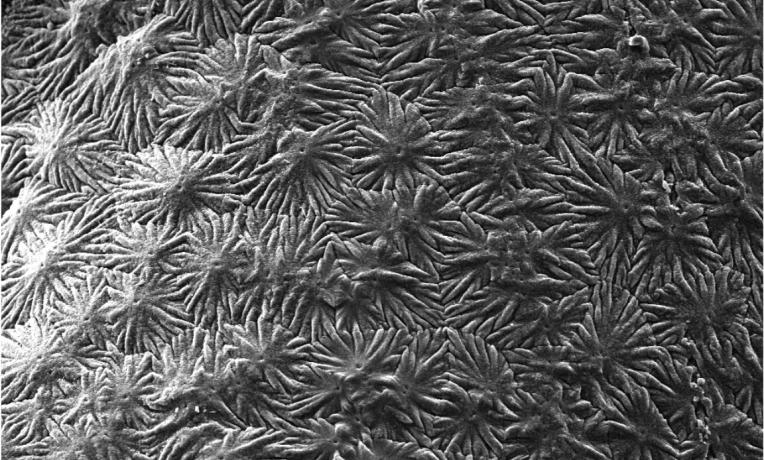
Enamel, located on the outer part of our teeth, is the hardest tissue in the body and enables our teeth to function for a large part of our lifetime despite biting forces, exposure to acidic foods and drinks and extreme temperatures. This remarkable performance results from its highly organised structure. However, unlike other tissues of the body, enamel cannot regenerate once it is lost, which can lead to pain and tooth loss. These problems affect more than 50 per cent of the world’s population and so finding ways to recreate enamel has long been a major need in dentistry.
Mata's novel approach allows creating materials with remarkable precision and order, which look and behave like dental enamel. The mechanism is based on a specific protein material that is able to trigger and guide the growth of apatite nanocrystals at multiple scales - similarly to how these crystals grow when dental enamel develops in our body. This structural organisation is critical for the outstanding physical properties exhibited by natural dental enamel.
Learning from nature
“A major goal in materials science is to learn from nature to develop useful materials based on the precise control of molecular building-blocks. The key discovery has been the possibility to exploit disordered proteins to control and guide the process of mineralisation at multiple scales. Through this, we have developed a technique to easily grow synthetic materials that emulate such hierarchically organised architecture over large areas and with the capacity to tune their properties”, explains Alvaro Mata, who has also received EU support through a Marie Curie Integration Grant.
The materials open up opportunities to treat and regenerate dental tissues and could be used for a wide variety of dental complications, including tooth decay or dentin hypersensitivity. Furthermore, Mata's control of the mineralisation process opens the possibility to create materials with properties that mimic different hard tissues beyond enamel such as bone and dentin. As such, the work has the potential to be used in a variety of applications in regenerative medicine. In addition, the study also provides insights into the role of protein disorder in human physiology and pathology.